A) It would increase.
B) It would decrease.
C) It would remain constant.
D) Cannot be determined.
E) None of these (A-D) .
G) C) and E)
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
What is the value of the reaction quotient,Q,for the voltaic cell constructed from the following two half-reactions when the Zn2+ concentration is 0.0103 M and the Ag+ concentration is 1.35 M? Zn2+(aq) + 2e- Zn(s) ; ° = -0.76 V Ag+(aq) + e- Ag(s) ; ° = 0.80 V
A) 177
B) 131
C) ![]()
D) ![]()
E) ![]()
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A cell is set up with copper and lead electrodes in contact with CuSO4(aq) and Pb(NO3) 2(aq) ,respectively,at 25°C.The standard reduction potentials are: Pb2+ + 2e- Pb ° = -0.13 V Cu2+ + 2e- Cu ° = +0.34 V If sulfuric acid is added to the Pb(NO3) 2 solution,forming a precipitate of PbSO4,the cell potential:
A) increases
B) decreases
C) is unchanged
D) can't tell what will happen
E) none of these
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A common car battery consists of six identical cells,each of which carries out the reaction: Pb + PbO2 + 2HSO4- + 2H+ 2PbSO4 + 2H2O
The value of 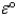 for such a cell is 2.039 V.Calculate G° at 25 °C for the reaction.
for such a cell is 2.039 V.Calculate G° at 25 °C for the reaction.
A) -196.7 kJ
B) -98.37 kJ
C) -393.5 kJ
D) -786.9 kJ
E) -590.2 kJ
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following reactions is possible at the anode of a galvanic cell?
A) Zn Zn2+ + 2e-
B) Zn2+ + 2e- Zn
C) Zn2+ + Cu Zn + Cu2+
D) Zn + Cu2+ Zn2+ + Cu
E) two of these
G) A) and C)
Correct Answer

verified
A
Correct Answer
verified
Multiple Choice
Which of the following is the strongest oxidizing agent? MnO4- + 4H+ + 3e- MnO2 + 2H2O = 1.68 V I2 + 2e- 2I- = 0.54 V Zn2+ + 2e- Zn =-0.76 V
A) MnO4-
B) I2
C) Zn2+
D) Zn
E) MnO2
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Nickel is electroplated from a NiSO4 solution.A constant current of 5.50 amp is applied by an external power supply.How long will it take to deposit 100.g of Ni? The atomic mass of Ni is 58.69.
A) 16.6 h
B) 8.30 h
C) 18.2 min
D) 55.4 s
E) 1.09 s
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which statement is always true of the cathode in an electrochemical cell?
A) It is considered the "negative" electrode.
B) It is considered the "positive" electrode.
C) Reduction occurs here.
D) Metal is plated out here.
E) Negative ions flow toward the cathode.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many electrons are transferred in the following reaction? 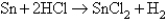
A) 0
B) 1
C) 2
D) 4
E) not enough information given
G) All of the above
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
In order to determine the identity of a particular lanthanide metal Sm,a voltaic cell is constructed at 25°C with the anode consisting of the lanthanide metal as the electrode immersed in a solution of 0.0819 M SmCl3,and the cathode consisting of a copper electrode immersed in a 1.00 M Cu(NO3) 2 solution.The two half-reactions are as follows: Sm(s) Sm3+(aq) + 3e-
Cu2+(aq) + 2e- Cu(s)
The potential measured across the cell is 2.67 V.What is the identity of the metal?

A) Ce
B) Eu
C) Dy
D) Sm
E) Gd
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which metal,Al or Ni could reduce Zn2+ to Zn(s) if placed in a Zn2+(aq) solution? Zn2+ + 2e- Zn = -0.76 V Al3+ + 3e- Al = -1.66 V Ni2+ + 2e- Ni =-0.23 V
A) Al
B) Ni
C) Both Al and Ni would work.
D) Neither Al nor Ni would work.
E) Cannot be determined.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In which direction do electrons flow in the external circuit?
A) left to right
B) right to left
C) no current flows;the cell is at equilibrium
D) cannot be determined.
E) none of these
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The following reaction occurs in aqueous acid solution: NO3- + I- IO3- + NO2 -In the balanced equation the coefficient of water is:
A) 1
B) 2
C) 3
D) 4
E) 5
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Balance the following oxidation-reduction reaction using the half-reaction method. Cr2O72- + I2 Cr3+ + IO3- In the balanced equation,the coefficient of water is:
A) 4
B) 17
C) 11
D) 7
E) 6
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For a certain reaction, H° = -74.4 kJ and S° = -227 J/K.If n = 3,calculate for the reaction at 25°C.
A) 0.0233 V
B) 0.491 V
C) 0.277 V
D) 0.0700 V
E) 0.237 V
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the galvanic cell shown below (the contents of each half-cell are written beneath each compartment) : 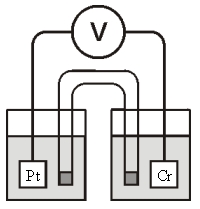 0.50 M Br2 0.20 M Cr3+ 0.10 M Br-
The standard reduction potentials are as follows:
Cr3+(aq) + 3e- Cr(s) =-0.727 V
Br2(aq) + 2e- 2Br-(aq) = +1.090 V
What is for this cell?
0.50 M Br2 0.20 M Cr3+ 0.10 M Br-
The standard reduction potentials are as follows:
Cr3+(aq) + 3e- Cr(s) =-0.727 V
Br2(aq) + 2e- 2Br-(aq) = +1.090 V
What is for this cell?
A) 1.817 V
B) 0.363 V
C) -0.363 V
D) 4.724 V
E) 1.316 V
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the cell potential at 25°C as read on the digital voltmeter?
A) 0.18 V
B) 2.58 V
C) 0.10 V
D) 0.59 V
E) 0.26 V
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What mass of chromium could be deposited by electrolysis of an aqueous solution of Cr2(SO4) 3 for 145 minutes using a constant current of 11.0 amperes?
A) 0.187 g
B) 154.7 g
C) 0.287 g
D) 25.8 g
E) 17.2 g
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider a galvanic cell based in the reaction Fe2+ + Cr2O72- Fe3+ + Cr3+ in acidic solution. -What is the coefficient of Fe3+ in the balanced equation?
A) 6
B) 2
C) 3
D) 4
E) none of these
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider an electrochemical cell with a zinc electrode immersed in 1.0 M Zn2+ and a nickel electrode immersed in 0.39 M Ni2+. Zn2+ + 2e- Zn ° = -0.76 V Ni2+ + 2e- Ni ° = -0.23 V Calculate for this cell.
A) 0.54 V
B) 0.52 V
C) 0.51 V
D) 0.53 V
E) 0.98 V
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 138
Related Exams